INTRODUCTION:NPM1 mutations (NPM1mut) occur in ∼30% of acute myeloid leukemia (AML) and frequently co-occur with mutations in other genes including those attributed to clonal hematopoiesis (CH) including DNMT3A and TET2, among others. CH mutations may persist beyond attaining NPM1mut-negative remission. Persistent CH may be associated with immunophenotypic alterations in myeloid progenitors detected by flow cytometry (FC) which poses an interpretive challenge in assessment of measurable residual disease (MRD) by FC. The aim of this study was to characterize the immunophenotypic alterations associated with persistent CH in the setting of NPM1mut clearance and to determine their possible clinical or biologic significance.
METHODS: The cohort included 67 consecutive patients (pts) with NPM1mut AML treated at our institution between 01/2017 and 11/2019. FC assessment for MRD was performed an eight-color panel using FACSCanto II instruments (BD Biosciences, San Diego, CA) with a sensitivity of 10-3 to 10-4. Whole bone marrow (BM) DNA was interrogated for mutations with an 81-gene myeloid next-generation sequencing (NGS) panel using an Illumina MiSeq sequencer (Illumina, Inc., San Diego, CA, USA) with a sensitivity: 1% variant allelic frequency (VAF).
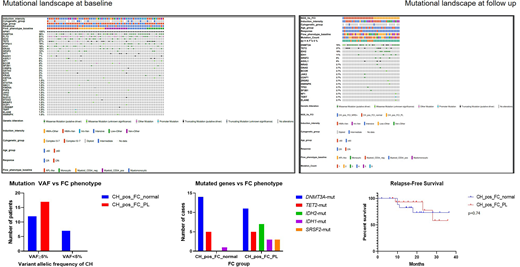
RESULTS: Pts included 26 men and 41 women with a median age of 64 (range, 19-84) years with newly diagnosed NPM1mut AML. AML blasts had the following immunophenotype at baseline: promyelocytic-like phenotype (CD34-, CD117+, HLA-DR-) in 18 (27%), aberrant myeloid CD34-/CD117+/HLA-DR+ in 15 (22%), aberrant myeloid CD34+ in 13 (19%), myelomonocytic in 11 (16%), and monocytic in 10 (15%) cases. All pts had additional co-mutations at baseline (Fig 1). The most frequently co-mutated genes were DNMT3A (58%) FLT3 (51%), TET2 (27%), IDH2 (24%), PTPN11 (19%), IDH1 (18%), NRAS (16%), and SRSF2 (12%). Pts were treated with intensive (35;52%) and non-intensive induction regimens (32; 48%) (Fig 1); 22 (33%) received an allogeneic hematopoietic stem cell transplant as post-remission consolidation. We compared FC and NGS results in follow-up samples in pts achieving NPM1mut negative remission with adequate data available for comparison (n=50). 13 (26%) pts cleared all mutations whereas 37 (74%) had persistent CH. The most common mutations in the setting of residual CH involved DNMT3A (70%), TET2 (27%), IDH2 (19%) and IDH1 (11%). Among 37 pts with residual CH, 19 (51%) had no phenotypic alterations detected by FC while 17 (49%) had myeloid progenitors with alterations in intensity of antigen expression (increased CD13, CD123, CD117 and/or decreased CD38) or deviation from normal maturation but not diagnostic for AML MRD (herein referred to as pre-leukemic (PL) phenotype); 1 sample was MRD+ by FC. Mutation VAF of ≥5% was significantly more common (p=0.008) in cases with FC PL+ (100%) vs cases with normal FC phenotype (63%). IDH2 and SRSF2 mutation were exclusively observed in PL+CH+ cases with the former being statistically significant when compared with the FC-normal group (p=0.016). PL phenotype by FC did not correlate with intensity of induction therapy (41% treated with intensive regimens vs 59% non-intensive). The CH+/PL+ cohort had more pts ≥60 yrs old (67%) but the difference was not significant. There was no significant association between PL+ and residual mutation count. Presence of PL+ phenotype was not associated with a shorter relapse-free survival (RFS) (median not reached for both groups).
CONCLUSIONS: Post-remission clonal hematopoiesis in the setting of NPM1mut clearance is common, and may result in immunophenotypic changes in myeloid progenitors, posing interpretive challenges for MRD assessment by FC. These alterations may be attributable to specific CH characteristics, such as IDH2 and SRSF2 mutations and VAF, but are not associated with a shorter RFS and thus should not be interpreted as residual AML or considered a high-risk attribute. Additional studies in other AML subtypes are warranted to further delineate these changes and their clinical significance.
DiNardo:Daiichi Sankyo: Consultancy, Honoraria, Research Funding; Notable Labs: Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy; Takeda: Honoraria; Jazz: Honoraria; ImmuneOnc: Honoraria; AbbVie: Consultancy, Honoraria, Research Funding; Syros: Honoraria; Agios: Consultancy, Honoraria, Research Funding; Calithera: Research Funding; MedImmune: Honoraria; Celgene: Consultancy, Honoraria, Research Funding. Short:AstraZeneca: Consultancy; Takeda Oncology: Consultancy, Honoraria, Research Funding; Amgen: Honoraria; Astellas: Research Funding. Kadia:JAZZ: Honoraria, Research Funding; Ascentage: Research Funding; Astra Zeneca: Research Funding; Incyte: Research Funding; Celgene: Research Funding; Novartis: Honoraria; Cyclacel: Research Funding; Genentech: Honoraria, Research Funding; Amgen: Research Funding; Abbvie: Honoraria, Research Funding; Cellenkos: Research Funding; Pulmotec: Research Funding; Astellas: Research Funding; BMS: Honoraria, Research Funding; Pfizer: Honoraria, Research Funding. Konopleva:Sanofi: Research Funding; Eli Lilly: Research Funding; AstraZeneca: Research Funding; Rafael Pharmaceutical: Research Funding; Amgen: Consultancy; F. Hoffmann La-Roche: Consultancy, Research Funding; AbbVie: Consultancy, Research Funding; Stemline Therapeutics: Consultancy, Research Funding; Kisoji: Consultancy; Reata Pharmaceutical Inc.;: Patents & Royalties: patents and royalties with patent US 7,795,305 B2 on CDDO-compounds and combination therapies, licensed to Reata Pharmaceutical; Ascentage: Research Funding; Calithera: Research Funding; Forty-Seven: Consultancy, Research Funding; Genentech: Consultancy, Research Funding; Cellectis: Research Funding; Agios: Research Funding; Ablynx: Research Funding. Kantarjian:Adaptive biotechnologies: Honoraria; Aptitute Health: Honoraria; BioAscend: Honoraria; Delta Fly: Honoraria; Janssen: Honoraria; Oxford Biomedical: Honoraria; Ascentage: Research Funding; Daiichi-Sankyo: Honoraria, Research Funding; BMS: Research Funding; Immunogen: Research Funding; Jazz: Research Funding; Novartis: Honoraria, Research Funding; Pfizer: Honoraria, Research Funding; Sanofi: Research Funding; Actinium: Honoraria, Membership on an entity's Board of Directors or advisory committees; Abbvie: Honoraria, Research Funding; Amgen: Honoraria, Research Funding. Ravandi:Macrogenics: Research Funding; Celgene: Consultancy, Honoraria; AstraZeneca: Consultancy, Honoraria; Xencor: Consultancy, Honoraria, Research Funding; Jazz Pharmaceuticals: Consultancy, Honoraria, Research Funding; Abbvie: Consultancy, Honoraria, Research Funding; Amgen: Consultancy, Honoraria, Research Funding; Astellas: Consultancy, Honoraria, Research Funding; BMS: Consultancy, Honoraria, Research Funding; Orsenix: Consultancy, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.